1. What makes snowflakes take shape?
Snowflakes begin forming high up in the sky when a water droplet freezes around a tiny particle (like a piece of dust), creating an ice crystal. As the crystal falls from the sky, water vapor freezes onto the crystal to create the six arms of the snowflake. Air and wind conditions as the snowflake falls (including temperature, humidity, wind speed and direction) determines its shape! That’s why no two snowflakes are exactly alike.

2. Why are all snowflakes symmetrical?
Amazing but true: even though every snowflake is different, they are all symmetrical. Do you know why? It’s because, when water freezes into ice or snow, water molecules align in a way that maximizes attractive forces and minimizes repulsive ones. Since water molecules are all the same size and shape, they form very ordered (and symmetrical!) arrangements when they crystallize.
Here is some more information about snowflakes, and a little chart which shows the different range of shapes.
.jpg)
Here is a fun craft and science project (in one!) for your child to make their own snowflakes, like the one pictured below:
.jpg)
3. What makes icicles pointy?

An icicle begins when a dangling water droplet starts to freeze before falling to the ground. In doing this, it forms a hollow tube of ice that continues to grow downwards. As water continues to drip down the outside of the icicle, its tip freezes faster than its base, which gives icicles the carrot shape we are all familiar with. Impurities in the water form ripples around their circumference; icicles made from pure water will be perfectly smooth.
Here’s a time-lapse video showing the amazing growth of icicles!
4. Why does ice float in water?

Ice floats because it is less dense than water. Density is a foundational concept in physics, which explains why boats float, rocks sink, and balloons fly away when you let them go! When water freezes into ice, it takes up more volume than when it does as a liquid.
As an experiment, see what happens if you put snow in a glass of water — does it float?
Here are some more fun density experiments you can try!
5. Why will your tongue stick to a cold metal pole?
We all know the classic “tongue stuck on a flagpole” trope, but why does it happen? Thermal conductivity. Thermal conductivity is a fancy way of saying that the metal will bring a tongue to its temperature much more quickly than your tongue will warm up the metal. Metal’s amazing ability to conduct heat is what will make a tongue freeze almost the moment it comes into contact with the frozen metal surface of, say, a flagpole.
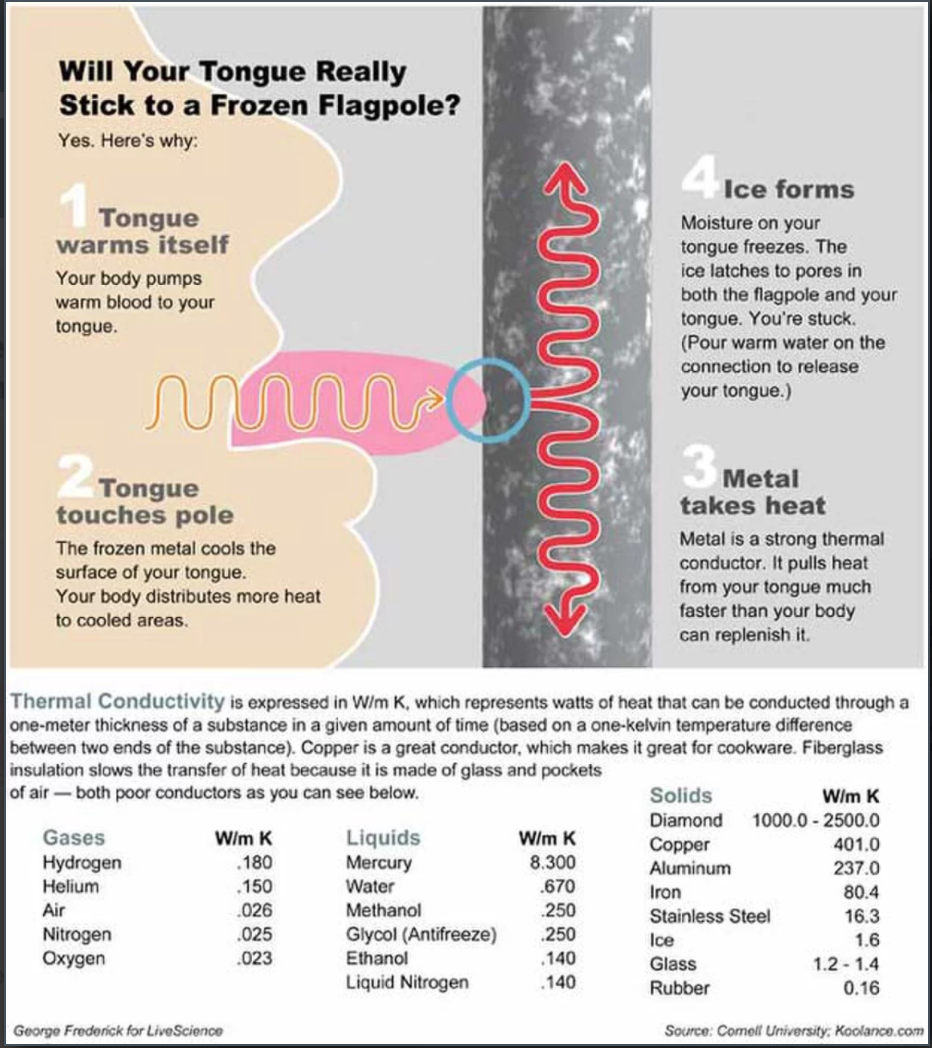
Here’s a fun (and MUCH safer!) way to experiment with water’s ability to quickly unfreeze and refreeze. In other words, what makes it appear “sticky”!
References:
- https://www.merriam-webster.com/words-at-play/the-less-lovely-side-of-snowflake
- http://earthsky.org/earth/how-do-snowflakes-get-their-shape
- https://www.scientificamerican.com/article/why-are-snowflakes-symmet/
- https://modernfarmer.com/2015/01/icicle-farming-real/
- https://www.theglobeandmail.com/technology/science/the-mysterious-and-cool-science-of-icicles/article23241318/
- http://chemistry.elmhurst.edu/vchembook/122Adensityice.html
- https://www.livescience.com/32237-will-your-tongue-really-stick-to-a-frozen-flagpole.html







